Since the start of the COVID-19 pandemic, more than 360,000,000 COVID-19 tests have been conducted in the United States alone, according to data from the COVID Tracking Project. With such unprecedented demand — not just across the nation, but around the world — it quickly became vital for U.S. healthcare providers to find new American suppliers of COVID-19 tests to reduce production and distribution lead times, increase test quality, and improve result accuracy.
Recognizing that their suppliers would not be able to scale to meet their testing requirements, the medical professionals at the Fox Chase Cancer Center, a research hospital in Pennsylvania, quickly found themselves in such a position.
“[As] the supply of swabs plummeted… We started having all these meetings about where we were going to get swabs,” said Dr. Eric M. Horwitz (MD, FABS, FASTRO), Chair of Radiation Oncology at Fox Chase.
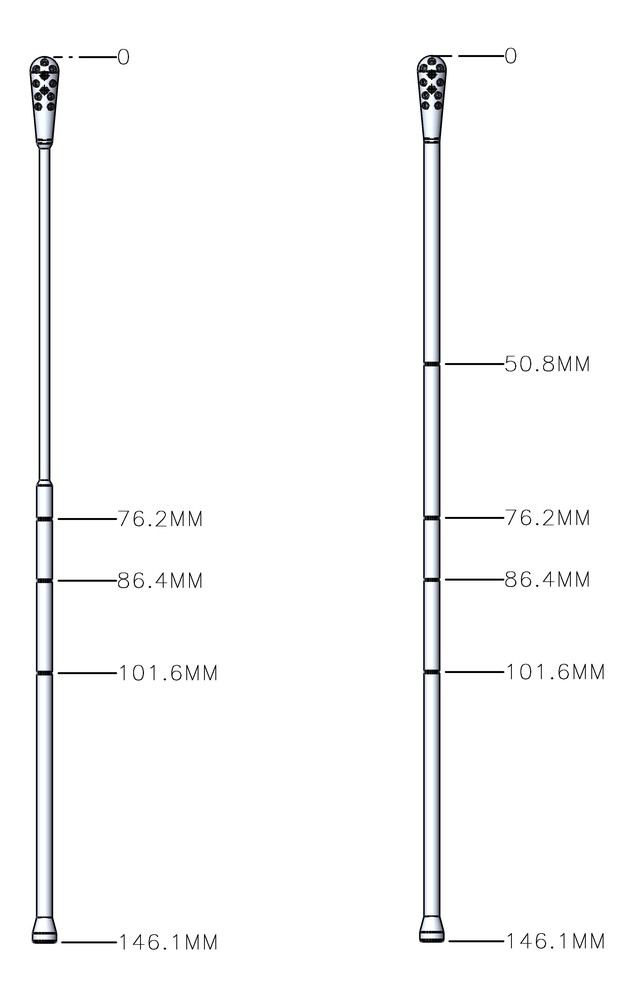
But Dr. Horwitz had an idea. His former patient, Joel Glickman, was the chairman of The Rodon Group, a local, third-generation plastic injection molding business. Dr. Horwitz explained the situation to Glickman, who quickly rallied the team at Rodon to develop and manufacture a new nasal and oral collection swab to exact medical specifications.
Rodon’s innovative fluid trap-tip design allowed for enhanced sample collection and release capabilities, making the testing process more comfortable for patients and more effective for medical professionals.

“We came up with a quick study to test people who were already getting swabbed. These patients would be swabbed twice, first with the one Fox Chase had been using and then with the new test swab,” Dr. Horwitz said. “It turned out that the one [The Rodon Group] designed actually worked even better than the one we had.”
Rodon’s swabs, which have already been FDA registered, are expected to receive ISO 13485 certification later this quarter. The company expects to produce 40-50 million swabs annually.
“We were proud to be able to support the staff at Fox Chase in their essential work,” said The Rodon Group CEO Michael Araten. “We believe it’s critically important, now more than ever, that healthcare devices can be made safely in the United States. We look forward to continuing to be a part of the ongoing effort to manufacture high-quality, American-made medical devices both through this pandemic and beyond.”
The Rodon Group, an ISO 9001:2015 certified, high-volume custom plastic injection molder, makes billions of parts each year in its 125,000 square foot Pennsylvania facility. To learn more about Rodon’s nasal testing swabs in their latest case study or to secure sample collection swabs for your testing needs, click here.