More than a million patients have been directly affected by 3D printing — and the numbers are set to grow.
We sat down to speak with technology advocate Lauralyn McDaniel to discuss additive manufacturing (AM) — technology that constructs 3D objects by adding layer on top of layer of material — in the medical field. She explained that the various scaffolds created are “the step between traditional 3D printing and full-on bioprinting, which involves actually printing living cells.”
In the past, doctors have had to rely on either tissue from cadavers or the patient’s own skin grafts. Now, she says, the scaffolds “help tissues to regenerate themselves, take advantage of the body’s own regenerative capabilities, and give the tissue a bridge on which to grow.”
“The same can be done with bones,” said McDaniel. “Particularly if you have a shattered bone, you can’t just set it and have it regrow — you need to put something in there. You can use scaffolds to regrow the bone rather than having to use metals and replacement parts to fuse it in. It can be done specifically to the patient’s need and size rather than forcing in off-the-shelf bone plate.”
In our conversation, we discuss how patents and FDA clearances are advancing 3D printing and how volunteers are playing a prominent role in AM medical.
Thomas Insights (TI): Additive manufacturing has been used since 1988. Why are we hearing so much more about it now?
Lauralyn McDaniel (LM): It started to really ramp up about eight years ago when patents started expiring on some of the primary processes. That meant there were more people making machines to do additive manufacturing.
One of the other reasons is that the processes have improved to the point where they’re faster. They’re more reliable. They’re more accurate. More materials are being developed to do this. And there are some large organizations that have invested heavily into the technology.
The U.S. government is highly interested and has made a lot of investment in the area. The number of standards that are now available for the processes, primarily metals and powder bed fusion, has grown dramatically.
And although they’ve kept it quiet for over 10 years, the medical device companies are using it as a production method for primarily orthopedic implants, although there are well over a hundred devices that have been cleared by the FDA in the U.S.
In the last four years, the number of softwares available to help optimize your additive manufacturing process from design tools to simulation tools to planning tools specific to the needs of additive manufacturing has quadrupled. Back in 2016, there were about 20 software packages, and now there are well over 150.
TI: Related to that, Materialise Mimics inPrint is the first software intended for 3D printing anatomical models for diagnostic use to receive FDA clearance. Why did it take so long for the software to get FDA clearance, and how do you think that will impact the industry?
LM: Between the FDA’s technical guidance and [hospital] groups setting their own best practices, it was recognized that anything to be used for diagnostic purposes should be using FDA-cleared software. That was in December 2017, so it didn’t take that long after that kind of recognition for Materialise to go, oh, yeah, we need to clear this. Since then, there’ve been two more softwares cleared.
TI: Collaboration was one of the top trends over the past year, according to a recent report. It was noted that this happened both among companies and through volunteers. How are volunteers shaping additive manufacturing?
LM: When you talk about volunteers, I’m specifically talking about the standards development work, which is generally consensus standards. That is volunteers of subject matter experts working together, donating their time, and it can be tedious and time-consuming work to do that. Setting a standard on a reliable way of doing something helps everyone so they don’t have to reinvent and find their own process and make mistakes.
We have the device manufacturers. We have the technology developers. We have the researchers. They all work together and all they all have special skills. In this area, when you’re talking about using additive manufacturing in medicine, it requires that combination of engineering and medicine and biology and technology to work together to address the challenges and to impact more patients with the benefits of 3D printing.
TI: Tell us about ASME’s AM Medical conference — who is the conference for, and what should they do to get the most out of the conference?
LM: The conference is first for those who are using the technology, and then second for those who are developing the technology.
And for the users; We’re talking to four major groups. The first is the medical device manufacturers who are using it, see it as a more efficient way to produce some of the structures they needed, the complexity they needed, enable some design of devices that can’t be achieved any other way.
And then you have the point-of-care manufacturers who use it in the clinical settings — those are the hospitals. They’re using a lot of anatomical models, but some of the leading hospitals like Mayo Clinic are starting to use it for developing patient-matched surgical guides. They’ll do the anatomical model, and then they’ll work with the surgeon, and the surgeon will say, ‘I want to cut here. I want to cut here,’ and then they’ll print a guide that they can fit onto the patient. It has the potential to dramatically cut the amount of time spent in the OR.
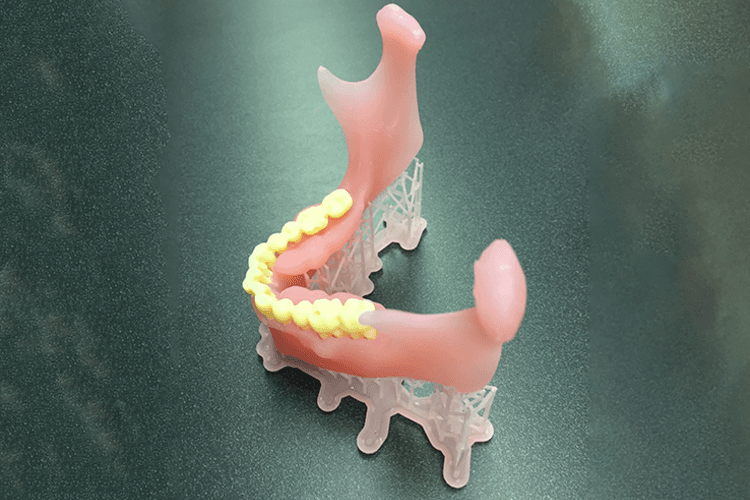
The third group is all those working in the dental applications area, so it includes the network of dental labs and dental offices across the country. And they have a unique advantage in that dentistry is always focused on patient-matched, patient-specific solutions — devices, instruments, that sort of thing. They’ve found that additive manufacturing or 3D printing is a more efficient way to do that. They can cut down weeks or even months on the time to develop something for a particular patient.
The final area, which is a growing area, is what we’re calling 3D biofabrication. It does include both the area of using advanced materials to develop scaffolds and structures to regenerate material as well as the actual printing of tissues.
The AM Medical Virtual Summit will take place May 27, 2020. It is hosted by the American Society of Mechanical Engineers (ASME).
This conversation has been lightly edited for clarity.